PerFix™ Plug
Plug and patch designed for use in a tension-free open inguinal hernia repair technique.


- Overview
- EIFU & Resources
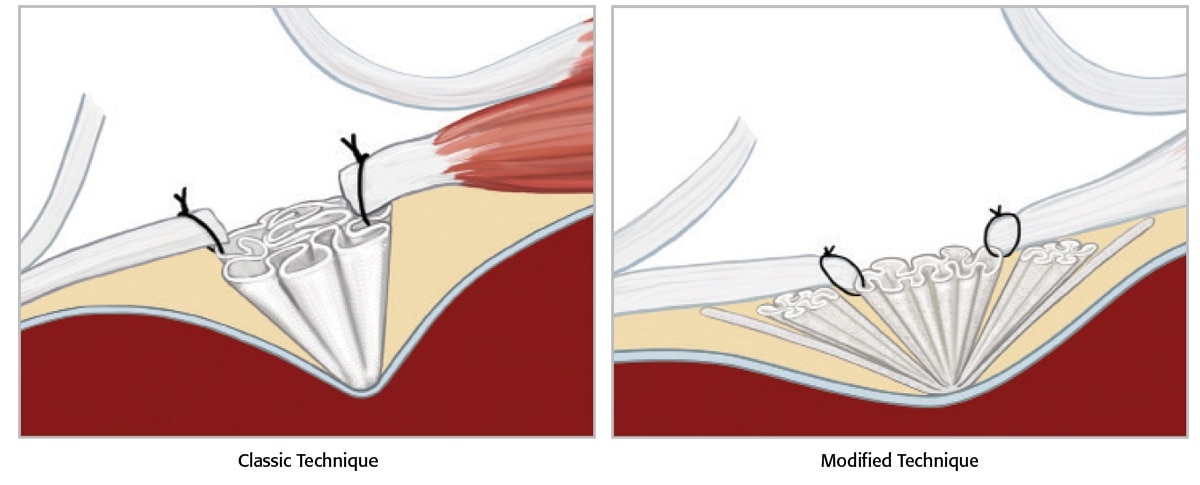
A quick and simple preperitoneal underlay Modified Technique for the repair of groin hernias
The PerFix™ Plug is ideal for use in a tension-free preperitoneal repair technique. With over five million implants worldwide, the Bard™ PerFix™ Plug is designed with pleated edges that conform readily to defects of various sizes and shapes. The monofilament polypropylene design ensures healthy tissue ingrowth.
- Entire operation can take 25 minutes or less2
- Minimal dissection is required
- 4-5cm incision1
- Local or epidural anesthesia can be used
- Accommodates all types of groin hernias
- Utilized in a tension-free repair technique
- Recurrence rates reported at 0.15%1
- Less than 0.5% chronic pain rate1
Please note, not all products, services or features of products and services may be available in your local area. Please check with your local BD representative.
Indications.
The PerFix™ Plug is indicated for use in the reinforcement of soft tissue where weakness exists, in the repair of inguinal and femoral hernias.
Contraindications.
- Do not use the PerFix™ Plug in infants, children, or pregnant women whereby future growth may be compromised by use of such mesh material.
- The use of this mesh has not been studied in breastfeeding or pregnant women.
- Literature reports that there may be a possibility for adhesion formation when polypropylene mesh is placed in direct contact with the bowel or viscera.
Warnings.
- The use of any permanent mesh or patch in a contaminated or infected wound could lead to fistula formation and/or extrusion of the mesh.
- If an infection develops, treat the infection aggressively. Consideration should be given regarding the need to remove the mesh. An unresolved infection may require removal of the device.
- If the unused mesh has been in contact with instruments or supplies used on a patient or contaminated with bodily fluids, discard with care to prevent risk of transmission of viral infections.
- To prevent recurrences when repairing hernias, the mesh should be sized with appropriate overlap for the size and location of the defect, taking into consideration any additional clinical factors applicable to the patient. Careful attention to mesh fixation placement and spacing will help prevent excessive tension or gap formation between the mesh and fascial tissue.
- This device is for single use only. Do not resterilize or reuse any portion of the PerFix™ Plug. Reuse, reprocessing, resterilization or repackaging may compromise the structural integrity and/or essential material and design characteristics that are critical to the overall performance of the device and may lead to device failure which may result in injury to the patient. Reuse, reprocessing, resterilization or repackaging may also create a risk of contamination of the device and/or cause patient infection or cross infection including, but not limited to, the transmission of infectious diseases from one patient to another. Contamination of the device may lead to injury, illness or death of the patient or end user.
- To avoid injury, careful attention is required if fixating the mesh in the presence of nerves, vessels or the spermatic cord. Fastener penetration into underlying tissue containing nerves or blood vessels may result in the need for medical/surgical intervention, cause serious injury or permanent impairment to a body structure.
- This device is supplied sterile. Inspect the packaging to be sure it is intact and undamaged prior to use.
- This device is not for the use of repair of pelvic organ prolapse.
- This device is not for the use of treatment of stress urinary incontinence.
Precautions.
- Care should be taken to ensure that the mesh is adequately fixated to uncompromised tissue. If necessary, additional fasteners and/or sutures should be used.
- Monofilament sutures are recommended to properly secure the mesh.
- Only physicians qualified in the appropriate surgical techniques should use this mesh.
- Please read all instructions prior to use.
Adverse Reactions.
Possible complications include, but are not limited to, seroma, adhesions, hematoma, pain, infection, inflammation, extrusion, erosion, mesh migration, fistula formation, allergic reaction and recurrence of the hernia or soft tissue defect.
Please consult package insert for more detailed safety information and instructions for use.
References
- Millikan KW, Doolas A. A Long-Term Evaluation of the Modified Mesh-Plug Hernioplasty in Over 2,000 Patients. Hernia. 2008 June; 12(3): 257-260.
2 Rutkow, I.M. : The Perfix plug repair for groin hernias. Surg Clinics of N. America, 2003; 83; 5: 1079-1098; 78; 6: 1007-1023
BD-60832
