RELATED PRODUCTS NOT AVAILABLE
true
Support
Customer Care & Ordering
866 979 9408, (905 288 6000 non toll free)
Contact Us
Thank you for contacting our sales team!
A sales representive will get in touch with you shortly.


- Overview
- Products & Accessories
- EIFU & Resources
The WavelinQ™ EndoAVF System is intended for the cutting and coagulation of blood vessel tissue in the peripheral vasculature for the creation of an arteriovenous fistula used for hemodialysis.
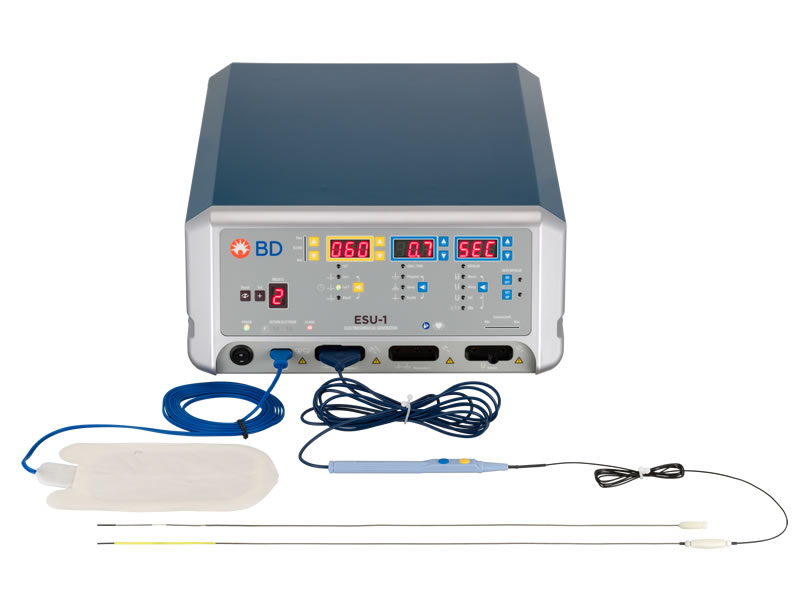
WavelinQ™ EndoAVF System
The WavelinQ™ EndoAVF System consists of two 4 French (4F) single-use, disposable, magnetic, hydrophilic coated catheters: a venous catheter and an arterial catheter. It is used with an Electrosurgical Unit (ESU) and Electrosurgical Pencil.

WavelinQ™ EndoAVF Arterial Catheter
The WavelinQ™ EndoAVF System arterial catheter contains a backstop for receiving the electrode. When placed in proximity, the magnets contained in each catheter attract, while aligning the electrode with the backstop. Two rotational indicators, present in each catheter, are used to accurately position the catheters. The catheters may be advanced and engaged in either a parallel (same direction) or antiparallel (opposite direction) configuration.

WavelinQ™ EndoAVF Venous Catheter
The WavelinQ™ EndoAVF System venous catheter is a flexible 4F magnetic catheter that contains a radiofrequency (RF) electrode for the delivery of RF energy, a yellow valve crosser on the distal end, and a cable and plug. The WavelinQ™ venous catheter connects via an Electrosurgical Pencil to an ESU for delivery of radiofrequency energy with standard grounding pads. RF energy can then be delivered through the electrode for cutting and/or coagulating tissue.
Literature
BD's collection of literature on industry and on our offerings gives you information you can use to continue striving for excellence.
Learn more
Events
BD supports the healthcare industry with market-leading products and services that aim to improve care while lowering costs. We host and take part in events that excel in advancing the world of health™.
Learn more
true
Please consult product labels and inserts for indications, contraindications, hazards, warnings, precautions and directions for use.
BD-23460
true