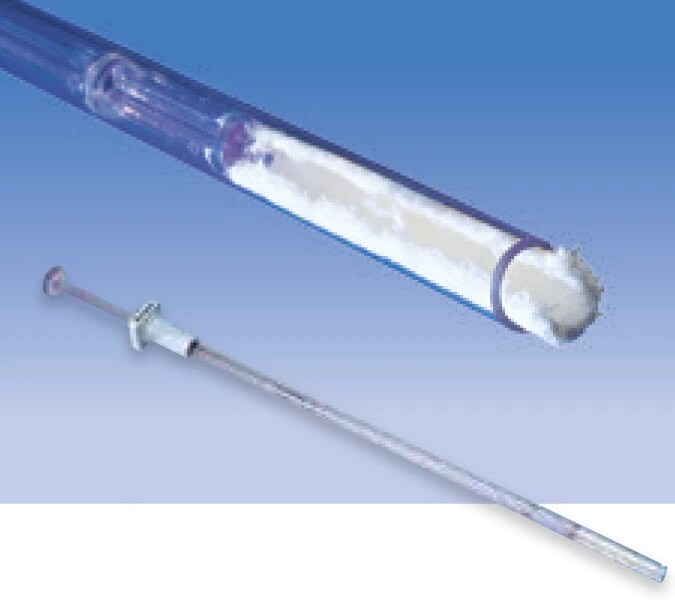
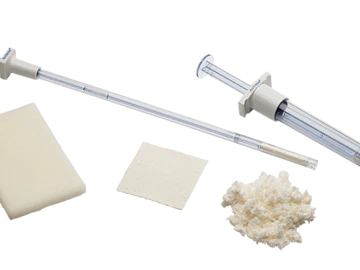
Avitene™ MCH is acceptable for use in all procedures where a topical hemostat is indicated. Its five versatile form factors provide an effective and simple solution for hemostasis.1
Avitene Sheets are composed of non-woven Avitene collagen fibrils in a sheet form which may be cut into any shape or size, and is highly versatile. Avitene Sheets are commonly used in intracranial and skull base surgery, e.g. tumor removal and neurovascular surgery.1
Avitene MCH is soft, fluffy, moldable form of microfibrillar collagen which is effective in controlling arterial hemorrhage. It conforms and adheres to irregular spaces and it’s easy to remove with gentle irrigation.1